Chemistry GK Practice Question and Answer
8 Q: Which of these metals is found in liquid state?
1957 061275cd493f02206607eb0af
61275cd493f02206607eb0af- 1Ironfalse
- 2Mercurytrue
- 3Silverfalse
- 4Othersfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 2. "Mercury"
Explanation :
Mercury is the only metal that is liquid at normal temperatures.
Q: The producers formed by the combustion of magnesium are -
1972 061275bc7dbe92e068b9111ff
61275bc7dbe92e068b9111ff- 1is acidicfalse
- 2is indifferentfalse
- 3alkalinetrue
- 4allfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 3. "alkaline"
Explanation :
Therefore, the correct option is (C) Magnesium ribbon on burning in air produces: 'Magnesium oxide, heat and light'.
Q: Which metal can be cut with a knife among sodium, lithium, calcium and nickel?
1804 061275c2293f02206607ea973
61275c2293f02206607ea973- 1Sodiumtrue
- 2Lithiumfalse
- 3calciumfalse
- 4allfalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 1. "Sodium"
Explanation :
Alkali metals (lithium, sodium, potassium) are so soft that they can be cut with a knife. The freshly cut surface is a shiny, silver colour, but this tarnishes quickly to a dull grey as the metal reacts with oxygen and water in the air.
Q: Which of the following elements is NOT a component of baking soda?
1135 164bfc857a2d4dcaf044da339
64bfc857a2d4dcaf044da339- 1Hydrogenfalse
- 2Calciumfalse
- 3Sodiumfalse
- 4Oxygentrue
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 4. "Oxygen"
Explanation :
1. Which of the following calcium elements is not a component of baking soda?
2. The chemical formula of baking soda is NaHCO3, which is composed of four elements sodium, hydrogen, carbon and oxygen.
3. Baking soda is an alkaline substance used as a leavening agent in baking.
Q: Which law was studied in the year 1787, in which it was said that the volume of a gas increases with its absolute temperature and if its absolute temperature decreases, then its volume will decrease?
1136 064b5204188d5e4f52dd2277b
64b5204188d5e4f52dd2277b- 1Boyle’s lawfalse
- 2Dalton’s lawfalse
- 3Avogadro’s lawfalse
- 4Charles's lawtrue
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 4. "Charles's law"
Explanation :
1. Charles's law (also known as volume law) is an experimental gas law.
2. The volume of a gas increases with its absolute temperature and as its absolute temperature decreases its volume also decreases.
3. Charles's law was studied in the year 1787.
Q: Identify a class of compounds that has a functional group –OH.
974 064b51d2e568e7ff594b01e05
64b51d2e568e7ff594b01e05- 1Alcoholtrue
- 2Ethanefalse
- 3Ketonefalse
- 4Aldehydefalse
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 1. "Alcohol"
Explanation :
1. That class of compounds, which contain a functional group –OH, is called alcohol.
2. Alcohols contain a hydrogen atom and a hydroxyl group (-OH) along with a carbon atom.
3. The hydroxyl group is a polar group, which makes the alcohol soluble in water.
Q: Hydrogen resembles the properties of which two groups of the periodic table?
1038 064a2b0fac7d7c7e0674a2640
64a2b0fac7d7c7e0674a2640- 1Group 2 and Group 17false
- 2Group 1 and Group 3false
- 3Group 1 and Group 17true
- 4Group 2 and Group 4false
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 3. "Group 1 and Group 17"
Explanation :
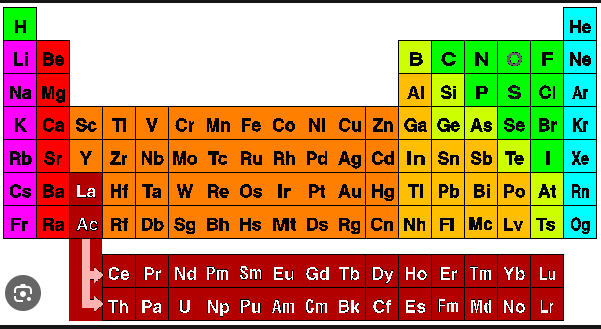
2. Following is some additional information about the properties of hydrogen.
- Hydrogen is a highly flammable gas.
-Hydrogen is a major component of water.
- Hydrogen can be used as fuel.
Hydrogen is used in the chemical industry to make many types of products.
Q: In 1845, which German chemist published a method for the synthesis of acetic acid, a natural product containing two carbon atoms?
984 0649aa86b1a612ce001f66c19
649aa86b1a612ce001f66c19- 1Friedrich Wohlerfalse
- 2Marcellin Berthelotfalse
- 3Joseph Gay-Lussacfalse
- 4Hermann Kolbetrue
- Show AnswerHide Answer
- Workspace
- SingleChoice
Answer : 4. "Hermann Kolbe"
Explanation :
1. In 1845, the German chemist who published a method for the synthesis of 'acetic acid', a natural product containing two carbon atoms, was Hermann Kolbe.
2. He created carbon disulfide with chlorine, and the end result was carbon tetrachloride.

